 Its function is to hydrolyze triglycerides (lavender) from lipoproteins into free fatty acids (FFA) and
monoglycerides (MG).
Its function is to hydrolyze triglycerides (lavender) from lipoproteins into free fatty acids (FFA) and
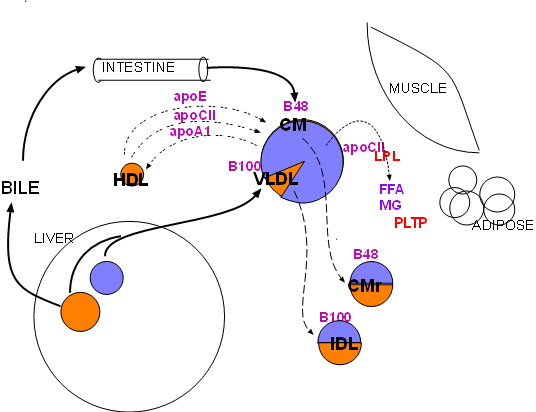
monoglycerides (MG).The largest circle in the diagram represents the liver. The small lavender circle within it represents stored triglycerides and the orange circle represents stored cholesterol (cholesterol esters). The mid-sized circle that has a triglyceride-rich (lavender) core represents the large lipoproteins that come from the intestines (chylomicrons) and from the liver (very low density lipoproteins, VLDL). The chylomicron has a specific apoprotein (B48) tightly bound to its coat; likewise the VLDL also has a similar bound apoprotein (B100). Both types of lipoprotein go through the same sequence of events -- hydrolysis of their triglycerides -- so the same particle represents them both. However, recall the size comparisons presented in the previous tutorial -- a chylomicron is significantly larger than a VLDL.
Lipoprotein lipase (LPL, red) is synthesized primarily by skeletal muscle and adipose tissue.
It is secreted into the interstitial fluid then diffuses to nearby capillaries. It becomes
attached to large, branched proteins (haparan sulfate
proteoglycans (HSPGs) bound to the inner capillary surface (not shown).
 Its function is to hydrolyze triglycerides (lavender) from lipoproteins into free fatty acids (FFA) and
monoglycerides (MG).
Its function is to hydrolyze triglycerides (lavender) from lipoproteins into free fatty acids (FFA) and
monoglycerides (MG).
However, lipoprotein lipase is inactive until it becomes bound to its cofactor, apoprotein CII (apoCII). Apoprotein CII is attached to VLDLs when they are secreted from the liver. Chylomicrons don't initially have this apoprotein but receive it (and apo E) from circulating high density lipoproteins (HDL)(see small-dashed arrows upper left). In exchange, the chylomicron transfers its apoprotein A1 to the HDL.
When the apo C-II on a triglyceride-rich lipoprotein contacts LPL on the capillary inner surface (note diagram) the lipolipase becomes activated and it hydrolyzes (dotted arrow) the triglyceride in the lipoprotein into free fatty acids(FFA) and monoglycerides (MG). The lipoprotein and attached enzyme can detach from the capillary wall and circulate; the hydrolysis continues as long as the enzyme remains attached.
The dashed arrows pointing from the triglyceride-rich lipoprotein to the two smaller circles indicates that lipoproteins shink as the lose their triglycerides. As this occurs excess coat material (cholesterol, phospholipids, apoproteins) is picked up by phospholipid transfer protein (PLTP). With the loss of apo C-II, during the decrease in size, hydrolysis will cease.
The smaller lipoproteins are called 'remnants'; the remnant of VLDL is also called intermediate density lipoprotein (IDL). Their cores have approximately equal amounts of cholesterol ester and triglyceride (note colors in diagram.)
The concentration of lipoprotein lipase in the above-mentioned tissues varies with the nutritional state at the time. This is best understood by considering blood glucose levels. After a meal (postprandial) a high glucose level stimulates the pancreas to release insulin into the blood. In addition to facilitating glucose uptake by cells, insulin also affects triglyceride metabolism.
The effect of insulin on adipocytes is to stimulate them to produce and secrete lipoprotein lipase. This facilitates the uptake of free fatty acids and monoglycerides into this tissue for conversion back to triglycerides for storage. Simultaneously, the effect of insulin on skeletal muscle is to inhibit the secretion of lipoprotein lipase. Overall this encourages triglyceride storage in adipose tissue rather than oxidation in muscles.
When fasting (low blood glucose), glucagon instead of insulin is the major hormone involved. The effect of glucagon on skeletal muscle is to stimulate the production and secretion of lipoprotein lipase This encourages free fatty acids to be oxidized for energy. Glucagon has no effect on adipose tissue.
Under stressful conditions, when adrenalin is circulating, both skeletal and cardiac muscles are stimulated to produce and secrete lipoprotein lipase. This supplements the glucose oxidation normally occurring under these circumstances.
Continue to Remnants
Return to home page